Recently, as the first author, Ph.D student Bi Fukun from School of Environment and Architecture published a paper titled “Chlorine-Coordinated Pd Single Atom Enhanced the Chlorine Resistance for Volatile Organic Compound Degradation: Mechanism Study” in Environmental Science & Technology. USST was the first unit; Zhao Zhenyuan, Yang Yang, Gao Weikang and professor Liu Ning were the co-authors; Dr. Zhang Xiaodong and professor Huang Yuandong were the corresponding authors.
Over the past few decades, as a kind of common gas pollutant, Volatile Organic Compounds,VOCs discharged from chemical industry, dyestuff, etc, are the important precursors of ozone and PM 2.5, causing serious harm to the humans and environment. The research reveals that volatile Organic Compunds react with strong oxidizing substances in the air with the effect of light to produce ozone and secondary organic aerosols. Secondary organic aerosols will absorb nitrate,sulfate, etc. In order to solve the current situation of air pollution, the “14th Five-year Plan” clearly proposed to strengthen the comprehensive treatment of volatile organic compounds. Therefore professor Huang Yuandong and Dr. Zhang Xiaodong led the team to carry out research in control and reduction of volatile organic compounds.
For the degradation of volatile organic compounds, catalytic oxidation is commonly used, that is, the volatile organic compounds are oxidized to carbon dioxide and water vapor at a low temperature due to catalyst. As far as this method is concerned, the most critical is to develop an efficient and economic catalyst. Noble metal supported catalysts, due to their excellent catalytic effects, are widely used in the low-temperature catalytic degradation of volatile organic compounds.
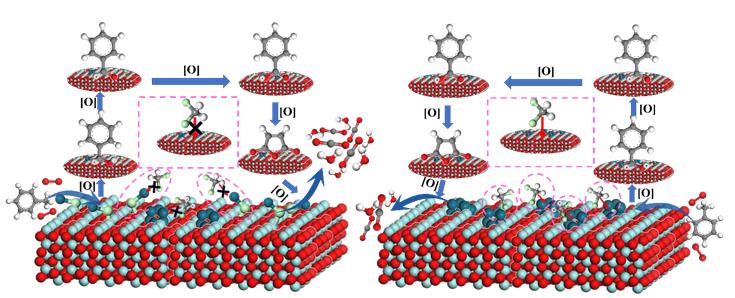
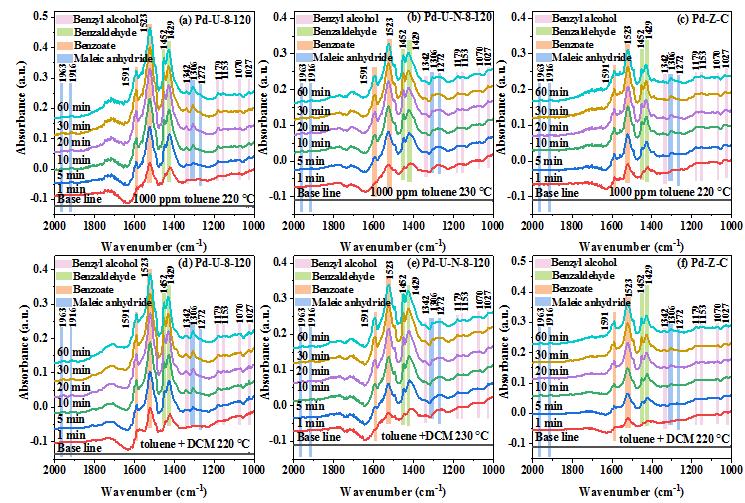
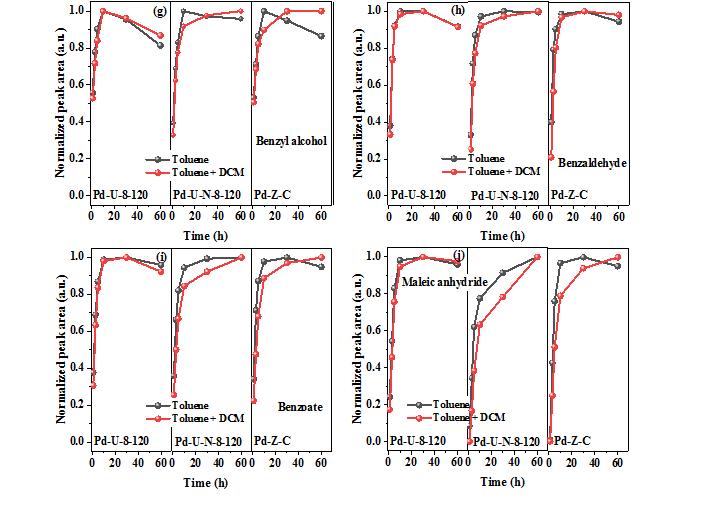
However, on the actual industrial emission conditions, the emission of volatile organic compounds is often accompanied by a small amount of chlorine containing volatile organic compounds. The presence of Pd1−Cl species is the key factor to enhance the chlorineresistance. The formation of Pd1−Cl species suppresses theadsorption of DCM on the active sites, enhances the exposure of more active sites, and reduces the accumulation ofintermediates on the surface of Pd@ZrO2-800. Furthermore, the monodispersed Pd atoms and high VO concentration in Pd@ZrO2-800 can improve the reactivity of gaseous oxygen molecule adsorption and dissociation, which accelerates the degradation of the intermediates to CO2 and H2O.42 However, without the presence of Pd1−Cl, the addition of DCM in the reaction system will lead to the adsorption of DCM on the catalyst surface, resulting in the occupation of active sites and further causing the accumulation of the degradation intermediates, which weaken the degradation of toluene to CO2 and H2O.
The research reveals that chlorine coordinated palladium monoatomic catalyst plays an important role in improving the chlorine resistance of supported noble metal catalyst in the degradation process, providing an important theoretical basis for developing a catalyst with high chlorine resistance for the degradation of volatile organic compounds. The research results are highly innovative.
Link to the paper: https://doi.org/10.1021/acs.est.2c06886


